Why are halogens not found free in nature?
Likewise, people ask, why do halogens not occur free in nature?
It's not as easy as you think. Fluorine is so reactive that we cannot find it free in nature. None of the halogens exist free in nature (unlike some of the metals such as gold and silver) because they are very reactive.
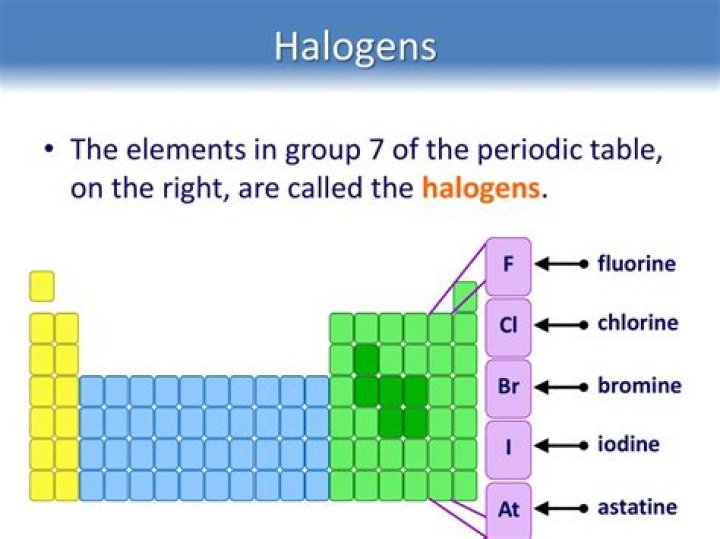
Subsequently, question is, are halogens found alone in nature? The halogens are chemical elements under the second-to-right column in the periodic table, also known as Group 17. The elements in the group are fluorine, chlorine, bromine, iodine, astatine, and ununseptium. They are rarely found alone in nature, except astatine, which is not found in nature.
Correspondingly, why are halogens volatile in nature?
The halogen molecules are more reactive.
What do halogens exist as?
All of the halogens exist as diatomic molecules. This means that the elements are made up of pairs of atoms that are chemically joined together (for example, fluorine exists as F 2, chlorine as Cl 2, bromine as Br 2 and iodine as I 2).
Related Question Answers
Why are metals not free in nature?
Alkaline metals and halogens do not occur in free state in nature. This is because they are unstable in their free state and react very quickly to gain stability. They make bonds with most of the elements. Therefore the halogens combine with other halogen atoms to form a covalent bond so as to get a stable octet.Why alkali metals do not occur free in nature?
Alkali metals are the chemical elements in group one of the periodic system. They are never found in nature uncombined because they are unstable and they react fast to other elements. They bond well with all elements except the noble gases.Why alkali metals and halogens do not occur free in nature?
Alkali metals and halogens do not occur free in nature because they are very reactive. They occur in a combined state.Do noble gases form compounds?
All noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Because of their high ionization energy and almost zero electron affinity, they were not expected to be reactive.Which of the following element is present in the group where bromine is present in modern periodic table?
The halogen elements are the six elements in Group 17 of the periodic table. Group 17 is the second column from the right in the periodic table and contains six elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (As), and tennessine (Ts).Why is bromine less volatile than chlorine?
Because a bromine atom has one more full shell than a chlorine atom, it has more shielding which means the attraction between the nucleus and an electron is weaker.Which memory is volatile in nature?
Volatile memory is computer memory that requires power to maintain the stored information. Most modern semiconductor volatile memory is either Static RAM (see SRAM) or dynamic RAM (see DRAM). SRAM retains its contents as long as the power is connected and is easy to interface to but uses six transistors per bit.What is the meaning of volatile nature?
A substance is said to be volatile if it boils at a low temperature, changing from the liquid to the gas phase. Substances that are gases at room temperature are extremely volatile: they have high volatility. They can only be seen as liquids when exposed to low temperatures or high pressures.Why do halogens become less volatile down the group?
As you go down the group, halogens become less volatile, as their shell size increases. This increases the Van der Waals forces as there are more electrons, hence increasing the force of attraction between molecules. This makes them less volatile.Which halogen is more volatile?
Boiling point of HF is highest due to H-bonding. For other halogen acids boiling point increase in the order HCl<HBr<HI. Therefore, most volatile (with lowest boiling point) is HCl.What is the heaviest element of the halogens?
astatineWhat properties do halogens have in common?
Halogens share many similar properties including:- They all form acids when combined with hydrogen.
- They are all fairly toxic.
- They readily combine with metals to form salts.
- They have seven valence electrons in their outer shell.
- They are highly reactive and electronegative.
Are halogens dangerous?
Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. Fluorine is one of the most reactive elements. It reacts with otherwise inert materials such as glass, and it forms compounds with the heavier noble gases. It is a corrosive and highly toxic gas.Which family of elements has only 1 valence electron?
A: Any element in group 1 has just one valence electron. Examples include hydrogen (H), lithium (Li), and sodium (Na). Any element in group 18 has eight valence electrons (except for helium, which has a total of just two electrons). Examples include neon (Ne), argon (Ar), and krypton (Kr).Which family of elements loses electrons most easily?
In particular, cesium (Cs) can give up its valence electron more easily than can lithium (Li). In fact, for the alkali metals (the elements in Group 1), the ease of giving up an electron varies as follows: Cs > Rb > K > Na > Li with Cs the most likely, and Li the least likely, to lose an electron.Are alkali metals brittle?
As with all metals, the alkali metals are malleable, ductile, and are good conductors of heat and electricity. The alkali metals are softer than most other metals.Why are halogens rarely found in native state?
All of the halogens are highly reactive, and will combine directly with almost all elements. Due to this high level of reactivity, the halogens are almost never found in pure form; rather, they have to be extracted.What is the symbol element with atomic number 74?
TungstenWhy do halogens have similar properties?
Atoms of group 7 elements all have seven electrons in their outer shell. This means that the halogens all have similar chemical properties .What Colour is chlorine?
greenWhat was the first halogen discovered?
Bromine was discovered in the 1820s by Antoine Jérôme Balard. Balard discovered bromine by passing chlorine gas through a sample of brine. He originally proposed the name muride for the new element, but the French Academy changed the element's name to bromine.Are halogens good conductors of electricity?
Like other nonmetals, halogens cannot conduct electricity or heat. Compared with most other elements, halogens have relatively low melting and boiling points.What is the difference between chlorine and fluorine?
Fluorine is a very reactive gas at room temperature. Chloride is a monovalent, stable, negative ion associated with other ions, most commonly metallic and in a solid state at room temperature. Chlorine is, as is fluorine, a highly reactive and toxic gas.Why is fluorine more reactive than chlorine?
The reactivity is the halogens ability to gain an electron, so number of electrons already in the atom plays a vital role. Chlorine has more electrons so repels a reacting electron with greater force than fluorine, making it less likely to react.Why are halogens kept in Group 17?
The halogens are located on the left of the noble gases on the periodic table. Because the halogen elements have seven valence electrons, they only require one additional electron to form a full octet. This characteristic makes them more reactive than other non-metal groups.What Colour is fluorine?
yellowWhich halogen is used in photography?
Silver chloride, silver bromide (AgBr) and silver iodide (AgI) are the three silver "halide" compounds used in photography. The halide elements include fluorine (F), chlorine (Cl), bromine (Br) and iodine (I).What Colour is bromine?
Physical and chemical propertiesFree bromine is a reddish brown liquid with an appreciable vapour pressure at room temperature. Bromine vapour is amber in colour.


