Where does Gibbs free energy come from?
Correspondingly, what is the meaning of Gibbs free energy?
Gibbs free energy is a measure of the potential for reversible or maximum work that may be done by a system at constant temperature and pressure. It is a thermodynamic property that was defined in 1876 by Josiah Willard Gibbs to predict whether a process will occur spontaneously at constant temperature and pressure.
Likewise, why Gibbs energy is negative? Explanation: Gibbs free energy is a derived quantity that blends together the two great driving forces in chemical and physical processes, namely enthalpy change and entropy change. If the free energy is negative, we are looking at changes in enthalpy and entropy that favour the process and it occurs spontaneously.
Herein, how do you get Gibbs free energy?
To get an overview of Gibbs energy and its general uses in chemistry. Gibbs free energy, denoted G, combines enthalpy and entropy into a single value. The change in free energy, ΔG, is equal to the sum of the enthalpy plus the product of the temperature and entropy of the system.
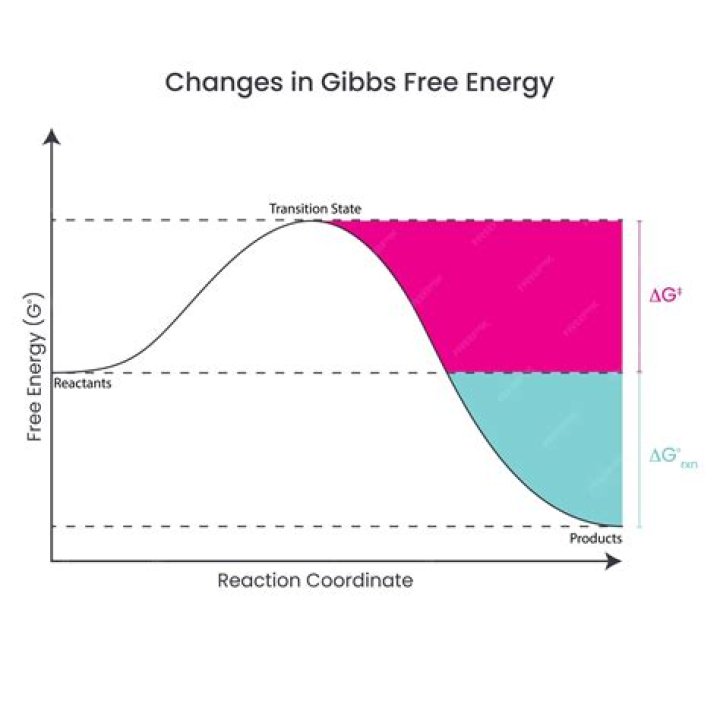
Why is Gibbs free energy at equilibrium?
At equilibrium, a system is in dynamical equilibrium. Both the forward and backward reactions occur at the same rate. If the Gibbs energy change for the forward reaction is G, then the Gibbs energy change is -G for the backward reaction. This is why Gibbs energy is zero at equilibrium.
Related Question Answers
Why is it called free energy?
Free Energy refers to the energy in a system that is free to do work i.e. the internal energy minus any energy that is unavailable to perform work. It's normally called the Gibbs energy more recently, though at my Uni it's often been refered to as the 'Gibbs Free Energy'.When Gibbs free energy is negative?
A negative ∆G means that the reactants, or initial state, have more free energy than the products, or final state. Exergonic reactions are also called spontaneous reactions, because they can occur without the addition of energy.Is Gibbs free energy a path function?
Answer and Explanation: Gibbs free energy (G) is a state function since it depends on enthalpy (H), absolute temperature (T) and entropy (S), all of which are stateWhat is an example of free energy?
For example, the energy for the maximum electrical work done by a battery as it discharges comes both from the decrease in its internal energy due to chemical reactions and from the heat TΔS it absorbs in order to keep its temperature constant, which is the ideal maximum heat that can be absorbed.How do you get free energy?
Gibbs free energy, denoted G, combines enthalpy and entropy into a single value. The change in free energy, ΔG, is equal to the sum of the enthalpy plus the product of the temperature and entropy of the system.What is the concept of free energy?
Free energy is that portion of any first-law energy that is available to perform thermodynamic work at constant temperature, i.e., work mediated by thermal energy. Free energy is subject to irreversible loss in the course of such work. The historically earlier Helmholtz free energy is defined as A = U − TS.What does it mean when Delta G is 0?
Unfavorable reactions have Delta G values that are positive (also called endergonic reactions). When the Delta G for a reaction is zero, a reaction is said to be at equilibrium. Equilibrium does NOT mean equal concentrations. If the Delta G is zero, there is no net change in A and B, as the system is at equilibrium.What is the unit for Gibbs free energy?
Gibbs free energy, denoted G, combines enthalpy and entropy into a single value. The change in free energy, ΔG, is equal to the sum of the enthalpy plus the product of the temperature and entropy of the system.Gibbs Energy in Equilibria.
| Keq | ΔGo(kcal/mole) |
|---|---|
| 1 | 0 |
| 101 | -1.36 |
| 102 | -2.73 |
| 103 | -4.09 |
Is Delta G 0 for elements?
No. Elements occur in different allotropes. ΔHof and ΔGof are defined to be zero at 298K, 1 bar for the lowest energy allotrope, with the exception that the values for white phosphorous are defined to be zero even though it is not the lowest energy allotrope.How does temperature affect Gibbs free energy?
Free Energy (G) can either increase or decrease for a reaction when the temperature increases. It depends on the entropy (S) change. The change in a quantity is represented by the Greek letter delta. Hence, when the temperature increases the numeric value of the free energy becomes larger.Is negative delta G spontaneous?
For a spontaneous reaction, the sign on Delta G must be negative. Gibbs free energy relates enthalpy, entropy and temperature. A spontaneous reaction will always occur when Delta H is negative and Delta S is positive, and a reaction will always be non-spontaneous when Delta H is positive and Delta S is negative.What is Delta G in biology?
Every chemical reaction involves a change in free energy, called delta G (∆G). Endergonic reactions require an input of energy; the ∆G for that reaction will be a positive value. Exergonic reactions release free energy; the ∆G for that reaction will be a negative value.What does Delta G mean?
Delta G is the symbol for spontaneity, and there are two factors which can affect it, enthalpy and entropy. Enthalpy - the heat content of a system at constant pressure. When delta G > 0 - It's a non-spontaneous reaction.What is Delta H and Delta S?
Delta S refers to the change of Entropy. And delta H refers to the change of enthalpy.What is r in Delta G equation?
In this equation: R = 8.314 J mol-1 K-1 or 0.008314 kJ mol-1 K-1. T is the temperature on the Kelvin scale. Top.What is Delta H?
In chemistry, the letter "H" represents the enthalpy of a system. Enthalpy refers to the sum of the internal energy of a system plus the product of the system's pressure and volume. The delta symbol is used to represent change. Therefore, delta H represents the change in enthalpy of a system in a reaction.What do you mean by Helmholtz free energy?
In thermodynamics, the Helmholtz free energy is a thermodynamic potential that measures the useful work obtainable from a closed thermodynamic system at a constant temperature and volume (isothermal, isochoric).What does a decrease in free energy mean?
If free energy decreases, the reaction can proceed. If the free energy increases, the reaction can't proceed. A reaction is favored if the free energy of the system decreases. A reaction is not favored if the free energy of the system increases.What does negative delta s mean?
A negative delta S would mean that the products have a lower entropy than the reactants, which is not spontaneous by itself.Can entropy be negative?
Entropy is the amount of disorder in a system. Negative entropy means that something is becoming less disordered. In order for something to become less disordered, energy must be used. The second law of thermodynamics states that the world as a whole is always in a state of positive entropy.Is Gibbs free energy always positive?
Keep in mind that the temperature in the Gibbs free energy equation is the Kelvin temperature, so it can only have a positive value.Gibbs Free Energy.
| ΔH | ΔS | ΔG |
|---|---|---|
| negative | positive | always negative |
| positive | positive | negative at higher temperatures, positive at lower temperatures |

