SN1 stands for S -SUBSTITUTION , N -NUCLEOPHILIC, 1- UNIMOLECULAR. SN2 stands for S -SUBSTITUTION , N -NUCLEOPHILIC, 2- BIMOLECULAR. If you want to have in-depth knowledge on SN1 & SN2 Mechanism you please go through the link cited below. Just so, what is meant by sn1 and sn2?
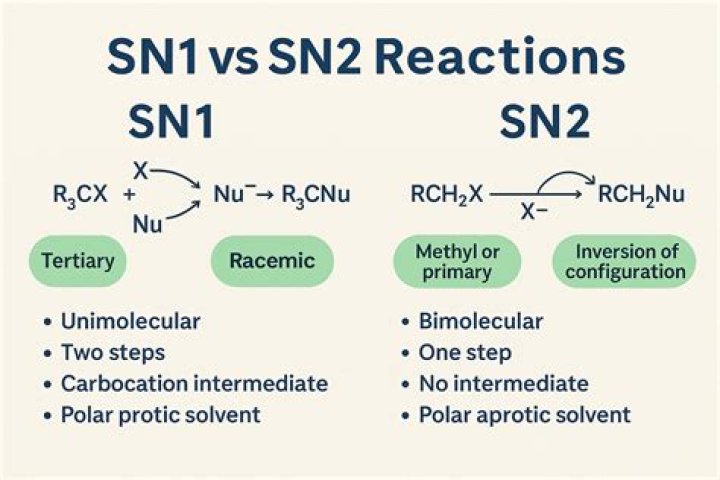
SN1 reactions are nucleophilic substitutions, involving a nucleophile replacing a leaving group (just like SN2). However: SN1 reactions are unimolecular: the rate of this reaction depends only on the concentration of one reactant. ? SN1 reactions happen in two steps: 1.
Also, why are sn1 and sn2 reactions important? SN1 reactions are important because, as far as we know, they describe a mechanism of organic reactivity, of chemical reactivity. And they describe a BOND-BREAKING PROCESS, as compared to SN2 reactions, which are bond-making processes with respect to the rate determining step.
Also to know is, what is sn1 and sn2 reaction with example?
SN2 and SN1 reactions are types of nucleophilic substitution reaction that often involve substitution of one nucleophile (such as OH) by another nucleophile.
Is sn1 or sn2 faster?
SN2 take place faster . its a one steped process . and SN 1 is two steped process in which first step that is formation of carbcation is slow and second step that is attack of nucleophile is fast .
Related Question Answers
What does sn2 mean?
SN2 Definition. The SN2 reaction - A Nucleophilic Substitution in which the Rate Determining Step involves 2 components. -SN2 reactions are bimolecular with simultaneous bond-making and bond-breaking steps. Why is sn1 faster than sn2?
5. For SN2, The Rate Of Reaction Increases Going From Tertiary To Secondary To Primary Alkyl Halides. For SN1 The Trend Is The Opposite. For the SN2, since steric hindrance increases as we go from primary to secondary to tertiary, the rate of reaction proceeds from primary (fastest) > secondary >> tertiary (slowest). Which is correct order of rates for sn1 for?
The rates of SN1 reactions decrease in the order tertiary > secondary > primary > > methyl. This trend is exactly the reverse of the trend observed in SN2 reactions. The relative reactivity of haloalkanes in SN2 reactions corresponds to the relative stability of carbocation intermediates that form during the reaction. Is Carbocation formed in sn2 reaction?
The stability of carbocation formation will determine if Sn1 or Sn2 reactions occur. In the second step, the electronegative nucleophile attacks the carbocation to form the product. The strength of nucleophiles used help to determine the reaction mechanism. Strong bases will almost always proceed to Sn2 mechanism. Is sn2 stereospecific?
The SN2 reaction is stereospecific. A stereospecific reaction is one in which different stereoisomers react to give different stereoisomers of the product. For example, if the substrate is an R enantiomer, a frontside nucleophilic attack results in retention of configuration, and the formation of the R enantiomer. Why is sn2 bimolecular?
Bimolecular reaction A bimolecular reaction, such as the SN2 reaction, is one in which two reactants take part in the transition state of the slow or rate-determining step of a reaction. For this reason, the concentrations of both the nucleophile and the alkyl halide are proportional to the observed SN2 reaction rate. Is hydrolysis sn1 or sn2?
Hydrolysis is a special type of nucleophilic substitution (SN1) where water acts as both nucleophile and a solvent molecule. Is sn2 second order?
2. The Rate Law Of The SN2 Is Second Order Overall. Note how the rate of the reaction is dependent on both the concentration of the nucleophile and that of the substrate. In other words, it's a second-order reaction. Why does sn1 result in Racemisation?
a) In SN1 reaction there is formation of carbocation intermediate in rate determining first step. So the nucleophile can attack from any side to carbocation. This give rise 50% product with D-configuration and 50% product with L configuration. Why is sn2 important?
The SN2 Reaction Is Incredibly Powerful And Can Be Used To Build A Large Number Of Functional Groups From Alkyl Halides. Note – some of these substitution reactions work better than others, especially on secondary carbons – depending on conditions, elimination reactions can start to compete when strong bases are used. Which sn2 reaction faster?
Explanation: SN2 reactions involve a backside nucleophilic attack on an electrophilic carbon. As a result, less steric congestion for this backside attack results in a faster reaction, meaning that SN2 reactions proceed fastest for primary carbons. Is Cl or Br a better leaving group?
like you said Br- is bigger than Cl- and can therefore better stabilize the negative charge, making it a better leaving group. What is the order of sn1 reaction?
In an SN1 reaction, the rate law is 1stt order. That is, the reaction rate depends on the concentration of only one component, the alkyl halide. Hence the term Substitution Nucleophilic 1st order. In an SN2 reaction, the rate law is 2nd order. Is Bromobenzene sn1 or sn2?
Bromobenzene does not react via SN1 or SN2 pathway because the structure of the ring does not allow for a backside attack in the case of SN2 or the formation of a carbocation in SN1. How do you tell if a nucleophile is strong or weak?
Nucleophilicity increases as the density of negative charge increases. An anion is always a better nucleophile than a neutral molecule, so the conjugate base is always a better nucleophile. A highly electronegative atom is a poor nucleophile because it is unwilling to share its electrons.