What is the main pattern for electronegativity?
Similarly, it is asked, what is the pattern for electronegativity?
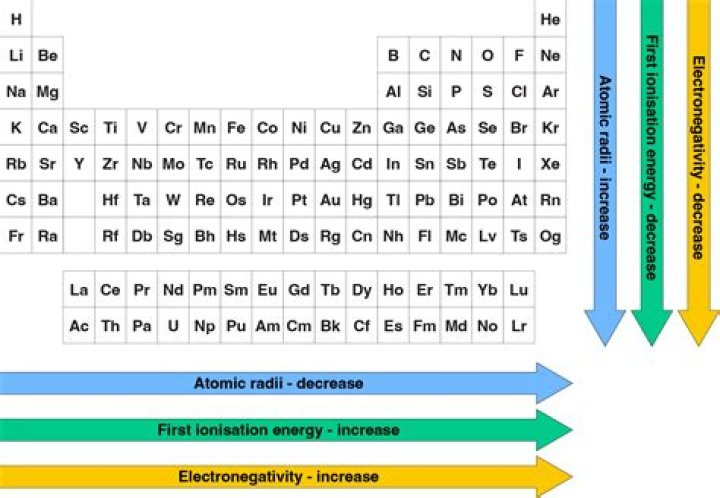
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
Similarly, what is the main pattern for electronegativity electron affinity and ionization energy? The trends of ionization energy, electron affinity, and electronegativity on the periodic table are the same. From left to right AND down to up on the table, the elements will have an increase in these trends. So if the ionization energy of an atom is high, the atom is more reluctant in giving up an electron.
Moreover, what is the proper order of electronegativity?
The order of electronegativities F > O > N > C is an important property that we will use to explain the chemical properties of organic compounds. Electronegativities decrease from top to bottom within a group of elements.
Is the pattern of electronegativity absolute or general?
E.Is the pattern of electronegativity absolute or general (always true or generally true)? Thepattern is generally true.
Related Question Answers
What group has the highest electronegativity?
VII A group is helogens and it is most electronegative group. It contains fluorine, chlorine, bromine, iodine and astatine. The most electronegative group of elements in periodic table is VIIA OR 17 group comprising elements i.e.,Flourine(F),chlorine(Cl),bromine (Br),iodine(I) ,astatine(At)..Which is more electronegative N or Br?
There are various scales, which draw on various parameters, of which the Pauling scale is the most famous. You will have to look up the Pauling scale yourself, but the O atom, first row, and rightmost on the table, will be most electronegative, followed by nitrogen and bromine.How does electronegativity increase?
Electronegativity increases as you move across the periodic table from left to right. This occurs due to a greater charge on the nucleus, causing the electron bonding pairs to be very attracted to atoms placed further right on the periodic table. Electronegativity decreases as you move down the periodic table.Why is fluorine more electronegative than chlorine?
Answer and Explanation:Fluorine is more electronegative than chlorine because fluorine is smaller and has its electrons closer to the positively charged nucleus.
How do you arrange elements in order of increasing electronegativity?
- Electronegativity increases left to right across a row in the periodic table e.g. C < N < O < F.
- Electronegativity decreases as you move down a group in the periodic table e.g. F > Cl > Br > I.
- F is the most electronegative element.
How do you compare electronegativity?
As a general rule: An atom's electronegativity gets higher as you move to the right in the periodic table. An atom's electronegativity gets higher as you move up in the periodic table. Thus, the atoms in the top right have the highest electronegativities and the atoms in the bottom left have the lowest ones.Which element has a higher electronegativity than arsenic?
Sorted by Electronegativity (Pauling)| Name | Sym | |
|---|---|---|
| 2.18 | Arsenic | As |
| 2.19 | Phosphorus | P |
| 2.2 | Hydrogen | H |
| 2.2 | Iridium | Ir |
Why Fluorine is most electronegative?
Fluorine is the most electronegative element because it has 5 electrons in it's 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons, so since Fluorine is so close to ideal electron configuration, the electrons are held very tightly to the nucleus.What 3 elements have the highest electronegativity?
In this scale neon has the highest electronegativity of all elements, followed by fluorine, helium, and oxygen.Which is more electronegative ch2 or CH?
CH≡ CH is more electronegative as compared to CH2≡ CH2 Explain.Which is more electronegative oxygen or chlorine?
Oxygen is more electronegative than Chlorine. Although when we move towards the right of the periodic table, electronegativity increases., but since the size of Chlorine is large, Consequently, the nucleus of oxygen attracts the electrons more than does the nucleus of chlorine.What element in the second period has the largest atomic radius group of answer choices?
LithiumWhich is the correct order of electronegativity f'n o p?
Answer: The correct order of electronegativities of N,O,F & P is F>O>N>P.Why is 2nd ionization energy higher?
An element's second ionization energy is the energy required to remove the outermost, or least bound, electron from a 1+ ion of the element. Because positive charge binds electrons more strongly, the second ionization energy of an element is always higher than the first.How do you find the second electron affinity?
Second Electron Affinity- X−(g)+e−→X−2(g)
- Og+e−→O−(g)1st EA = -142 kJ mol−1.
- O−g+e−→O2−(g)2nd EA = +844 kJ mol−1.
Why does the trend of electron affinity and electronegativity is same in a period?
Eea generally increases across a period (row) in the periodic table, due to the filling of the valence shell of the atom. However, this trend applies only to Group-1 atoms. Electron affinity follows the trend of electronegativity: fluorine (F) has a higher electron affinity than oxygen (O), and so on.What can we learn about Radius ionization and electronegativity from periodic trends?
1 Expert Answer. Atomic Radius: Going down the periodic table, each period has higher atomic radius because each period represents an additional electron shell being added, which means the atoms must grow larger. Electronegativity: Electronegativity is the ability of an element to attract electrons.Why does electronegativity increase across a period?
It is no wonder the electron pair gets dragged so far towards the chlorine that ions are formed. Electronegativity increases across a period because the number of charges on the nucleus increases. That attracts the bonding pair of electrons more strongly.Which has more negative electron affinity O or O?
Actually, since O− is negatively-charged, it would be more difficult to accept an electron, and significant coulombic repulsion would make the electron affinity of O− more positive than that of O (which is negative).Why do noble gases have no electronegativity?
Electronegativity is the atom's ability to attract electrons within a bond. Since noble gases already have a full valence shell of electrons and are stable, they have no need for attracting electrons so no, they do not have electronegativity.Is electronegativity and ionization energy the same?
Re: Ionization Energy vs. Electronegativity. Ionization energy is the amount of energy it takes to remove an electron from an atom while electronegativity is the atom's ability to attract an electron.Why does the atomic radius decrease as electrons are added to a shell?
Atomic radius decreases across a period because valence electrons are being added to the same energy level at the same time the nucleus is increasing in protons. The increase in nuclear charge attracts the electrons more strongly, pulling them closer to the nucleus.Which ion has the largest radius?
franciumWhere is the lowest electronegativity found?
Electronegativity increases moving from the bottom left-hand corner of the periodic table toward the upper right-hand corner. The element with the lowest electronegativity value is francium, which has an electronegativity of 0.7. This value uses the Pauling scale to measure electronegativity.Which apex atom has highest electronegativity?
The most electronegative element is Fluorine with a score of 4.0 (the highest possible.) Across from Fluorine we also have N and O with high electronegativities.Is the pattern of atomic radius absolute or general?
6. Is the pattern of atomic radius absolute or general (always true or generally true)?Activity – Trends in Atomic Radius.
| Li 145 | K 220 |
|---|---|
| Be 112 | Ca |
| B 85 | 1pm = |
| C | 1/1000000000000 |
| N 65 | of a metre |
What appears to be the trend in electronegativity as you move from left to right in a row?
The electronegativity of atoms increases as you move from left to right across a period in the periodic table. This is because as you go from left to right across a period, the nuclear charge is increasing faster than the electron shielding, so the attraction that the atoms have for the valence electrons increases.What is the periodic trend for electronegativity quizlet?
Electronegativity increases as you move from left to right across a period because the number of charges on the nucleus increases. As you move down a group (column), electronegativity decreases (as you move up, it increases) due to the distance between the nucleus and the valence electron shell.Which of the following elements has the highest electronegativity answers?
The most eletronegative elements are to the right of the periodic table. The electronegativities increase as you move right. Fluorine and Chlorine are the highest. Fluorine is the answer.What is the general trend of atomic radius?
Periodic TrendThe atomic radius of atoms generally decreases from left to right across a period. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. Within a period, protons are added to the nucleus as electrons are being added to the same principal energy level.