What is the difference between organic and inorganic solvent?
In respect to this, what is organic and inorganic solvent?
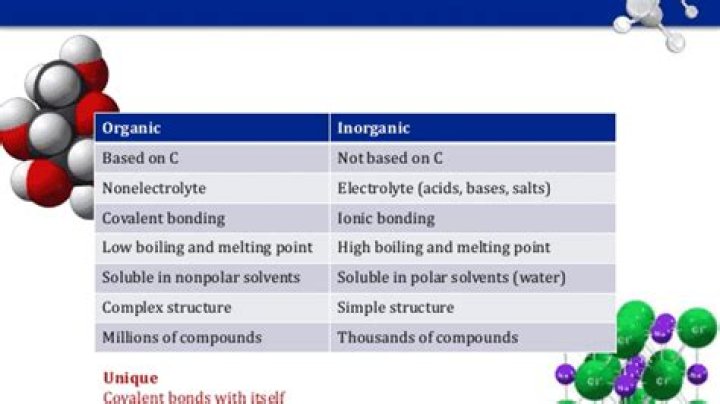
Water, the simplest and most abundant solvent on earth is classed as an inorganic solvent as its chemical structure does not contain carbon. Organic solvents (containing carbon) are classified by their chemical structure, of which there are three main types; Hydrocarbon solvents (aliphatic and aromatic)
One may also ask, what is meant by organic solvent? Organic solvents are carbon-based substances capable of dissolving or dispersing one or more other substances. Organic solvents can be carcinogens, reproductive hazards, and neurotoxins. Carcinogenic organic solvents include benzene, carbon tetrachloride, and trichloroethylene.
Secondly, what is the main difference between organic and inorganic chemistry?
The main difference is in the presence of a carbon atom; organic compounds will contain a carbon atom (and often a hydrogen atom, to form hydrocarbons), while almost all inorganic compounds do not contain either of those two atoms. While most inorganic compounds do not contain carbon, there are a few that do.
What is the most important inorganic solvent in nature?
water
Related Question Answers
What are 2 types of solvents?
Solvents can be broadly classified into two categories: polar and non-polar. A special case is mercury, whose solutions are known as amalgams; also, other metal solutions exist which are liquid at room temperature. Generally, the dielectric constant of the solvent provides a rough measure of a solvent's polarity.What are examples of organic solvents?
Examples of organic solvents- aromatic compounds, eg benzene and toluene.
- alcohols, eg methanol.
- esters and ethers.
- ketones, eg acetone.
- amines.
- nitrated and halogenated hydrocarbons.
Is baking soda organic or inorganic?
The chemical formula of baking soda is NaHCO3 ie sodium bicarbonate which is basically an inorganic compound. Thus baking soda is an inorganic compound.What is the most common inorganic solvent in the body?
WaterWhat are examples of solvent cleaners?
Solvent-based cleaners remain popular in industrial applications due to their cleaning power; they remove thick, baked on oils, dirt, containments, solder flux, and greases. Some examples of strong cleaning solvents are acetone, methyl ethyl ketone, toluene, nPB, and trichloroethylene (TCE).Is carbon monoxide organic or inorganic?
Compounds of carbon are classified as organic when carbon is bound to hydrogen. Carbon compounds such as carbides (e.g., silicon carbide [SiC2]), some carbonates (e.g., calcium carbonate [CaCO3]), some cyanides (e.g., sodium cyanide [NaCN]), graphite, carbon dioxide, and carbon monoxide are classified as inorganic.Is water inorganic?
The most general classification of chemical compounds is that of inorganic and organic substances. Inorganic substances generally do not contain a carbon atom, whereas organic substances contain several. Water is definitely an inorganic compound (dihydrogen oxide) and methyl alcohol is definitely an organic compound.Is kerosene organic or inorganic?
Though its use as an illuminant has greatly diminished, kerosene is still used extensively throughout Chemically, kerosene is a mixture of hydrocarbons. The chemical composition depends on its source, but it usually consists of about 10 different hydrocarbons, each containing 10 to 16 carbon atoms per molecule.What are the 5 main organic compounds?
Among the numerous types of organic compounds, four major categories are found in all living things: carbohydrates, lipids, proteins, and nucleic acids.What are 5 examples of organic compounds?
There are five major categories of organic compounds involved in normal physiology of the body:- Carbohydrates.
- Lipids.
- Proteins.
- Nucleic acids.
- High-energy compounds.
What are the four types of inorganic compounds?
In general, there are four groups of inorganic compound types. They are divided into bases, acids, salts, and water.Is sugar organic or inorganic?
The chemical formula for sugar is C6H12O6. The compound contains carbon, hydrogen, and oxygen. Sugar is processed from sugar cane, a plant. All living organisms contain carbon-based compounds, making them organic.Why is organic chemistry so hard?
One of the reasons many students find Organic chemistry so difficult is because Organic chemistry is a very demanding course. In Organic chemistry, students are tasked with learning a large number of reactions, nomenclature, and molecular theory, but we make this easy (How it Works).What makes something inorganic?
Inorganic molecules do not contain carbon-hydrogen bonds. While carbon can still appear in inorganic molecules, such as in carbon dioxide, it lacks the additional hydrogens that make it organic. However, they can still have covalent bonds, such as in water molecules or oxygen gas.Is protein organic or inorganic?
Carbohydrates, lipids, proteins and vitamins have carbon in their structure, making them organic. Water and minerals do not, so they are inorganic.Is inorganic chemistry harder than organic?
Organic Chemistry is harder than Inorganic Chemistry and therefore you would need extra efforts and more investment of time to learn the concepts and gain expertise. Understanding the concept is the key to master Organic Chemistry and you should invest more time in learning the foundations.What is organic and inorganic?
The word "organic" means something very different in chemistry than it does when you're talking about produce and food. inorganic compounds is that organic compounds always contain carbon while most inorganic compounds do not contain carbon. Also, nearly all organic compounds contain carbon-hydrogen or C-H bonds.Is water an organic solvents?
Because water is the biological solvent, most biological organic molecules, in order to maintain water-solubility, contain one or more charged functional groups.What characteristics does organic solvents have?
Organic solvents can be categorized by differences in molecular structure for each compound. High volatility, low boiling point, a colorless and clear liquid, and low molecular weight are physical properties typical of organic solvents. Examples of organic solvents include carbon tetrachloride and acetone.Is petrol an organic solvent?
Organic solvents include alcohols, ethers, ketones, aromatics, straight-chain alkanes, and common petroleum production.What are the types of solvents?
There are 3 types of solvents commonly encountered: nonpolar, polar aprotic, and polar protic.Is solvent dangerous?
Solvents, their vapours and mists have various effects on human health. Many of them have a narcotic effect, causing fatigue, dizziness and intoxication. High doses may lead to unconsciousness and death. Exposure to large doses of solvents may slow down reaction- time and affect rational judgement.Is acetone an organic solvent?
Acetone is a universal colorless, volatile, flammable organic solvent with the chemical formula (CH3)2CO. Also known as propanone or dimethyl ketone, it is the smallest and simplest ketone. Moreover, it is an industrial solvent. Acetone is an excellent solvent because it can dissolve other substances.Can be found where solvents are used?
Where are solvents used? Ink manufacture • Pharmaceutical manufacture • Paint manufacture Page 2 Solvents are also found in many products including: • cleaning and degreasing materials; • paint removers; • paints, lacquers and varnishes; • adhesives; • inks and ink removers; • pesticides; and • toiletries.Why lipid is soluble in organic solvent?
In general, neutral lipids are soluble in organic solvents and are not soluble in water. Some lipid compounds, however, contain polar groups which, along with the hydrophobic part, impart an amphiphilic character to the molecule, thus favoring the formation of micelles from these compounds.What are the common health hazards of organic solvents?
The principal health effects most typically associated with organic solvent exposure include nervous system damage (central and peripheral), kidney and liver damage, adverse reproductive effects such as sperm changes and infertility, skin lesions, and cancer.How organic and inorganic affect our body?
Those nutrients that contain carbon are called organic while those that do not contain carbon are called inorganic. Nutrients are substances required by the body to perform its basic functions. Since the human body does not synthesize nutrients, they must be obtained from the diet, making them essential.Why is water an inorganic solvent?
Water is an inorganic compound, a solvent .It does not possess any carbon in its molecular structure, hence not organic.


