An optically active compounds exists in two isomeric forms that rotate the plane polarized light in opposite directions. They are called optical isomers and the phenomena is called optical isomerism. The optical rotatory power of two isomers are equal in magnitude. but opposite in direction. Likewise, people ask, what is optical isomerism with example?
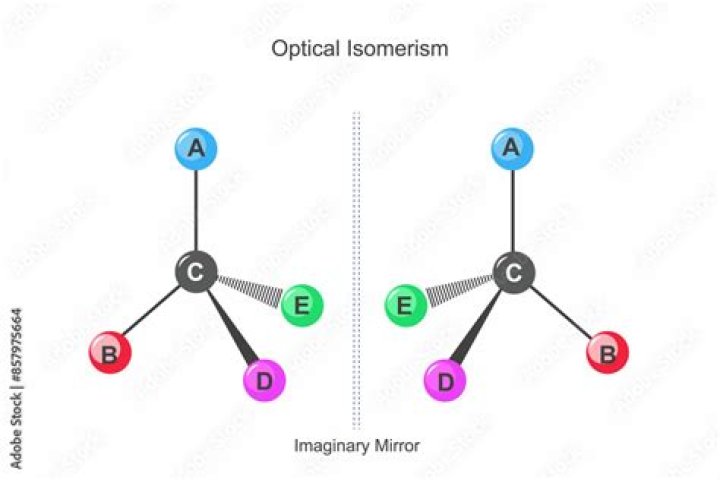
Optical isomers are compounds that are nonsuperimposable mirror images of each other. If the arrangement in space makes the two isomers nonsuperimposable mirror images of each other, we call them optical isomers or enantiomers. An example is the amino acid alanine.
Subsequently, question is, what are optical isomers Class 12? Hint: Optical isomers are molecules that differ from each other in their behavior towards plane-polarized light. They have different three-dimensional arrangements of the same atoms/groups in a molecule.
Then, how do you identify an optical isomer?
The "Plane of Symmetry Method" to Determine Optical Isomers
In other words, one looks for the existence of a plane of symmetry within the coordinate compound. If a plane of symmetry exists, then no optical isomers exist. On the other hand, if there is no plane of symmetry, the coordinate compound has optical isomers.
What are the types of optical isomers?
Optical isomerism
- Two compounds with the same brute formula are called isomers.
- There are different types of isomers: homomers, stereoisomers, constitutional isomers.
- In particular a stereocenter or stereogenic center is any point in a molecule bearing groups, such that an interchanging of any two groups leads to a stereoisomer.
Related Question Answers
What is the difference between enantiomers and optical isomers?
Optical isomers are two compounds which contain the same number and kinds of atoms, and bonds (i.e., the connectivity between atoms is the same), and different spatial arrangements of the atoms, but which have non-superimposable mirror images. Each non-superimposable mirror image structure is called an enantiomer. Which one will show optical isomerism?
2-hydroxypropanoic acid has a central carbon atom, which is linked to four different groups. This carbon atom is called chiral carbon atom and the molecule is called chiral molecule. Hence, 2-hydroxypropanoic acid shows optical isomerism. What can make a difference in optical isomers?
With optical isomerism, there is no difference in connectivity and no double bonds. The isomerism is to do with the arrangement of the atoms in space. It arises through the presence of a Chiral Centre. Optical isomers are Non Superimposable Mirror Images of each other; a set of optical isomers are called enantiomers. Why do optical isomers always come in pairs?
And optical isomers are isomers that are mirror images of each other. Like your hands, they come in pairs and are not superimposable. Well, these isomers got their name from how they rotate plane polarized light, or light waves that vibrate in only one direction. What do you mean by optical activity?
Optical activity, the ability of a substance to rotate the plane of polarization of a beam of light that is passed through it. The rotation is assigned a positive value if it is clockwise with respect to an observer facing the light source, negative if counterclockwise. Why is optical isomerism important in drug design?
Drug isomerism has opened a new era of drug development. Currently, knowledge of isomerism has helped us in introducing safer and more effective drug alternatives of the newer as well as existing drugs. Many existing drugs have gone chiral switch i.e., switching from racemic mixture to one of its isomers. How do you write an optical isomer?
For example, an enantiomer of alanine (amino acid) which rotates the plane of polarized light in clockwise and anti-clockwise direction can be written as (+) alanine and. How do you know if a compound shows optical isomerism?
Optical isomerism is exhibited by all species that rotate ppl in either clockwise or anticlockwise direction. This is checked by fact ( unsymmetrical molecules rotate ppl in either direction). What causes optical activity?
Theory. Optical activity occurs due to molecules dissolved in a fluid or due to the fluid itself only if the molecules are one of two (or more) stereoisomers; this is known as an enantiomer. Can tetrahedral have optical isomers?
In general, when dealing with a tetrahedral molecule that has 4 different ligands, optical isomers will exist most of the time. What is the criteria for optical activity in a coordination compound?
To check if a coordination compound is optically active, just see if if the mirror image of the compound overlaps with itself (for the given isomer), if it doesn't, the compound is optically active. How do you know if compounds are optically active?
To decide whether a compound should be optically active, we look for evidence that the molecules are chiral. The instrument with which optically active compounds are studied is a polarimeter, shown in the figure below. Imagine a horizontal line that passes through the zero of a coordinate system. How do you calculate total optical isomers?
Total number of optical isomers=2n−1. Number of enantiomers=2n−1−2(n−1)/2. Is optical isomerism a type of Stereoisomerism?
In stereoisomerism, the atoms making up the isomers are joined up in the same order, but still manage to have a different spatial arrangement. Optical isomerism is one form of stereoisomerism. Why optical isomers? Optical isomers are named like this because of their effect on plane polarised light. How many optical isomers does fructose have?
8 optical isomers
How many optical isomers are possible?
1 Answer. In principle there are 2n optical isomers for an organic molecule with n chiral centres. Of course that's in principle. Are all optical isomers optically active?
Optical isomers also have no axis of symmetry, which means that there is no line that bisects the compound such that the left half is a mirror image of the right half. Optical activity is the interaction of these enantiomers with plane-polarized light. Are diastereomers optical isomers?
8: Diastereomers differ at one or more atom. These molecules are not mirror images and they are not superimposable. They are optical isomers because they have the same connectivity between atoms but a different arrangement of substituent groups. What is D and L nomenclature?
The d/l system (named after Latin dexter and laevus, right and left) names molecules by relating them to the molecule glyceraldehyde. One example is the chiral amino acid alanine, which has two optical isomers, and they are labeled according to which isomer of glyceraldehyde they come from. Are meso compounds optical isomers?
Optical isomers are molecules with chiral centres that differ in the three-dimensional arrangement of their bonds. Hence, a meso compound is a stereoisomer of its optically active isomers. When you draw the two mirror image isomers of a meso compound, you are just looking at different views of the same compound. What are enantiomers 12?
Enantiomers are chiral molecules that are mirror images of one another. Furthermore, the molecules are non-superimposable on one another. This means that the molecules cannot be placed on top of one another and give the same molecule. Chiral molecules with one or more stereocenters can be enantiomers. What is optical activity Class 12?
(a) Optical activity is the property of optically active compounds to rotate the plane of plane-polarized light. Optically active compounds form nonsuperimposable mirror images. Optically active compounds do not show any symmetry such as symmetry about a point, about a line, and a plane. What are optical isomers used for?
Optical isomers can rotate the plane of plane-polarised light. When a solution is viewed from above: the (–) enantiomer rotates the plane anticlockwise (to the left) the (+) enantiomer rotates the plane clockwise (to the right). How many optical isomers does threonine have?
four possible
What does Epimer mean?
Epimer: One of a pair of stereoisomers that differ in the absolute configuration of a single stereocenter. When the molecule has only one stereocenter then the epimers are enantiomers. When the molecule has two or more stereocenters then the epimers are diastereomers.