Temperature has a direct effect on whether a substance exists as a solid, liquid or gas. Generally, increasing the temperature turns solids into liquids and liquids into gases; reducing it turns gases into liquids and liquids into solids. Furthermore, how is temperature related to changes in the state of matter?
Physical conditions like temperature and pressure affect state of matter. When thermal energy is added to a substance, its temperature increases, which can change its state from solid to liquid (melting), liquid to gas (vaporization), or solid to gas (sublimation).
Subsequently, question is, what is the effect of temperature on liquid state? As the temperature of a liquid increases, the molecules move faster thereby increasing the liquid's kinetic energy. Furthermore, the higher the temperature of the liquid, the lower the viscosity since an increase in kinetic energy reduces the forces of intermolecular attraction.
Similarly, it is asked, how does temperature affect solid liquid and gas?
Note how temperature effects the motion of the atoms or molecules in a liquid. As the temperature of a solid, liquid or gas increases, the particles move more rapidly. As the temperature falls, the particles slow down. If a liquid is heated sufficiently, it forms a gas.
At what temperature will a solid melt?
The melting point of ice is 0°C. The melting point of a solid is the same as the freezing point of the liquid. At that temperature, the solid and liquid states of the substance are in equilibrium.
Related Question Answers
Can Matter change its state?
All matter exists as solids, liquids, or gases. Matter can change from one state to another if heated or cooled. If ice (a solid) is heated it changes to water (a liquid). This change is called MELTING. Which property of ice gets changed on heating?
Answer: The properties of ice – melting and freezing are involved in its change during heating and cooling. Heating of ice leads to melting of ice. When ice is heated and temperature rises, thermal energy from heat is absorbed by the ice. Which state of matter has the lowest temperature?
Solid matter
How does temperature and pressure affect the state of matter?
If the pressure above a liquid is increased sufficiently, the liquid forms a solid. Physical conditions like temperature and pressure affect state of matter. The pressure law states that for a constant volume of gas in a sealed container the temperature of the gas is directly proportional to its pressure. What happens during state change?
During a change in state the heat energy is used to change the bonding between the molecules. In the case of melting, added energy is used to break the bonds between the molecules. Below is a picture of solid ice melting into liquid water. What is heat short answer?
Heat is the transfer of kinetic energy from one medium or object to another, or from an energy source to a medium or object. This is the amount of heat required to raise the temperature of one pound of pure liquid water by one degree Fahrenheit. How does temperature affect a solid?
Temperature has a direct effect on whether a substance exists as a solid, liquid or gas. Generally, increasing the temperature turns solids into liquids and liquids into gases; reducing it turns gases into liquids and liquids into solids. Which state of matter is most affected by temperature change?
Gases
Which has more pressure liquid or gas?
While the pressure exerted by a liquid or solid may be larger, the pressure of a gas is harder to escape. Slightly increasing the container size will dissipate most if not all of the pressure exerted by a liquid or solid. Gasses will continue exerting pressure as the container size increases. How do you know if an element is a solid liquid or gas?
Physical States — Melting Points, Boiling Points, and Densities. In the periodic table above, black squares indicate elements which are solids at room temperature (about 22ºC)*, those in blue squares are liquids at room temperature, and those in red squares are gases at room temperature. Can a solid turn into a gas?
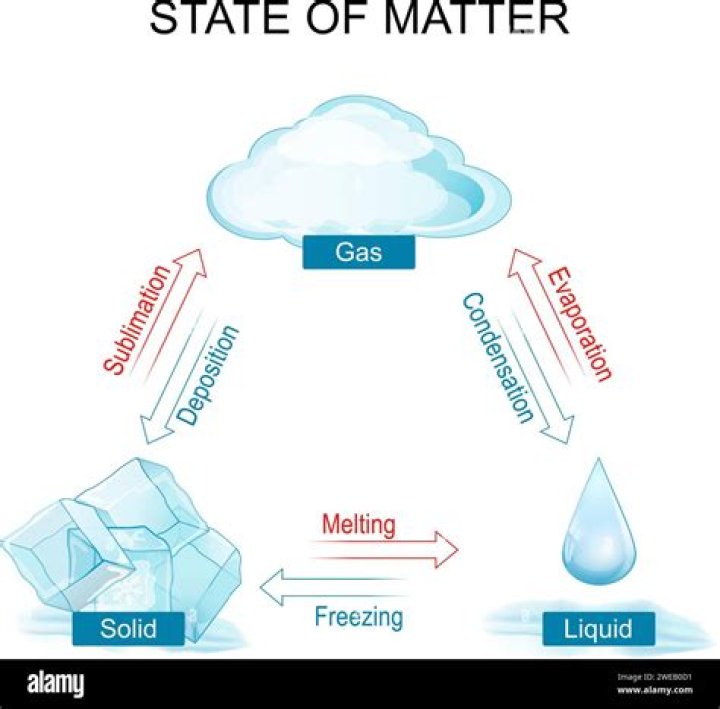
The process of changing a solid into a gas is called sublimation. An example of this process at normal atmospheric pressure is carbon dioxide (or dry ice) that will convert straight from the solid into a gaseous state. Is it possible that there are more than three states of matter?
Yes, there are many more states of matter that scientists have discovered, but only 3 are taught to middle school students. 2 of these are added to the "Big 3" states: plasma and Bose-Einstein condensate (BEC). Plasma is probably the most common state in the universe - not Earth. Plasma is essentially ionized gas. How does temperature affect gas?
1) If the volume of a container is increased, the temperature increases. 2) If the volume of a container is decreased, the temperature decreases. This means that the volume of a gas is directly proportional to its temperature. If you heat a gas you give the molecules more energy so they move faster. Does pressure decrease with temperature?
PRESSURE AND TEMPERATURE RELATIONSHIP. In a closed system where volume is held constant, there is a direct relationship between Pressure and Temperature. For example, when the pressure increases then the temperature also increases. When the pressure decreases, then the temperature decreases. What happens when a liquid is heated?
As the liquid is heated the the kinetic energy of the molecules increase and they liquid starts to expand. When the temperature reaches the boiling point of the liquid it starts boiling the phase changes from liquid to gaseous. What is it called when a solid turns into a gas?
For example, a solid may become a liquid. This phase change is called melting. When a solid changes into a gas, it is called sublimation. When a gas changes into a liquid, it is called condensation. When a liquid changes into gas, it is called evaporation. Does oil volume change with temperature?
For example, honey and oil tend to flow better at higher temperatures. In general, the liquids tend to expand when their temperature increases. For example, the same mass of boiling water occupies more volume at 100 degrees Celsius than at 20 degrees Celsius. Therefore, increasing temperature decreases density. How does temperature affect pressure?
The pressure law states that for a constant volume of gas in a sealed container the temperature of the gas is directly proportional to its pressure. This can be easily understood by visualising the particles of gas in the container moving with a greater energy when the temperature is increased. Why does temperature decrease with pressure?
For example, when the pressure increases then the temperature also increases. When the pressure decreases, then the temperature decreases. Because there is less mass in the can with a constant volume, the pressure will decrease. This pressure decrease in the can results in a temperature decrease. Does weight change with temperature?
Yes. If you have absolutely identical objects that have the same weight exactly when they are at the same temperature, then when one object is heated, it will weigh more. Temperature differences means that there is a different amount of kinetic energy in the motion of the atoms of the two bodies. Why does viscosity decrease with temperature?
The increase in temperature causes the kinetic or thermal energy to increase and the molecules become more mobile. The attractive binding energy is reduced and therefore the viscosity is reduced. Does temperature affect blood pressure?
Blood pressure generally is higher in the winter and lower in the summer. That's because low temperatures cause your blood vessels to narrow — which increases blood pressure because more pressure is needed to force blood through your narrowed veins and arteries. Does temperature affect flow rate?
In temperature programming, the flow rate will decrease as the column temperature increases. This decrease is due to the increased viscosity of the carrier gas at higher temperature. Does water volume increase with temperature?
An increase in temperature caused the water molecules to gain energy and move more rapidly, which resulted in water molecules that are farther apart and an increase in water volume. When water is heated, it expands, or increases in volume. When water increases in volume, it becomes less dense.